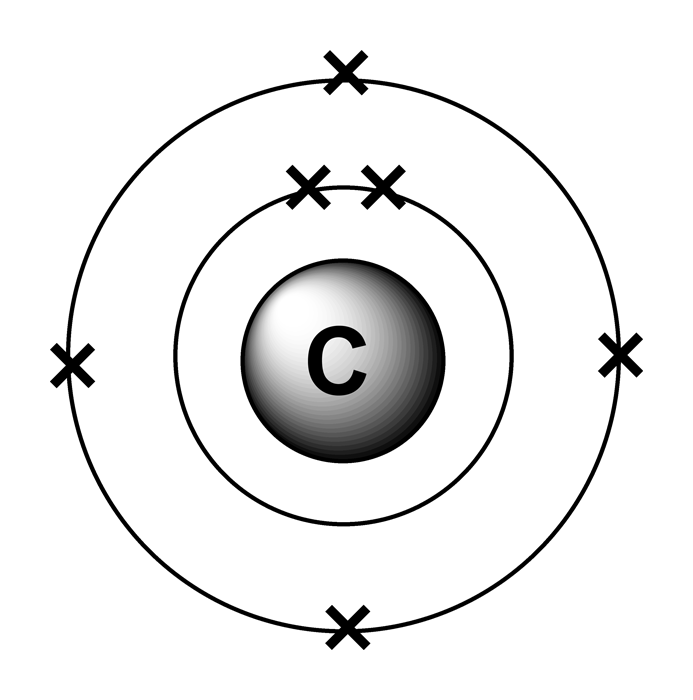
For example, in the sodium atom the highest n-value is 3. For the representative elements (columns 1, 2, and 13-18 of the Periodic Table), the core electrons are all electrons with an n-value lower than the maximum n-value in the electron configuration. For example, represents the 1 s 2 2 s 2 2 p 6 electron configuration of neon ( Z = 10), so the electron configuration of sodium, with Z = 11, which is 1 s 2 2 s 2 2 p 6 3 s 1, is written as 3 s 1Įlectrons in filled inner orbitals are closer to the nucleus and more tightly bound to it, and therefore they are rarely involved in chemical reactions. In practice, chemists simplify the notation by using a bracketed noble gas symbol to represent the configuration of the noble gas from the preceding row because all the orbitals in a noble gas are filled. Figure 2.1.1 tells us that the next lowest energy orbital is 2 s, so the orbital diagram for lithium isĪs we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner subshells. We know that the 1 s orbital can hold two of the electrons with their spins paired. The next element is lithium, with Z = 3 and three electrons in the neutral atom. Otherwise, our configuration would violate the Pauli principle. Written as 1 s 2, where the superscript 2 implies the pairing of spins. The orbital diagram for the helium atom is therefore From the Pauli exclusion principle, we know that an orbital can contain two electrons with opposite spin, so we place the second electron in the same orbital as the first but pointing down, so that the electrons are paired. We place one electron in the orbital that is lowest in energy, the 1 s orbital. Unless there is a reason to show the empty higher energy orbitals, these are often omitted in an orbital diagram: Figure 2.1.1), and the electron configuration is written as 1 s 1 and read as “one-s-one.”Ī neutral helium atom, with an atomic number of 2 ( Z = 2), has two electrons. Some authors express the orbital diagram horizontally (removing the implicit energy axis and the colon symbol): Here is a schematic orbital diagram for a hydrogen atom in its ground state: A filled orbital is indicated by ↑↓, in which the electron spins are said to be paired. We use the orbital energy diagram of Figure 2.1.1, recognizing that each orbital can hold two electrons, one with spin up ↑, corresponding to m s = +½, which is arbitrarily written first, and one with spin down ↓, corresponding to m s = −½. First we determine the number of electrons in the atom then we add electrons one at a time to the lowest-energy orbital available without violating the Pauli principle. We construct the periodic table by following the aufbau principle (from German, meaning “building up”). Similarly, the atoms of all group 7 elements have similar chemical properties and reactions to each other because all of them have seven electrons in their outer shell.\) The atoms of all group 1 elements have similar chemical properties and reactions because they all have one electron in their outer shell. The electronic configurations of the elements in group 1: 
So, elements in the same group have similar chemical properties because they have the same number of electrons in their outer shell. When atoms collide and react, it is the outer electrons that meet and interact. The electronic configurations of atoms help explain the properties of elements and the structure of the periodic table. has an atomic number of (2 + 8 + 1) = 11Įlectronic configurations and properties of elements.The electronic configuration of sodium (2.8.1) shows that sodium, Na: the number of electrons in all shells of an element is represented in the periodic table as the element's atomic number.the number of electrons in the outermost shell of an element is represented in the periodic table as the group number that element is situated in.the number of circles in the electronic configuration of an element is represented in the periodic table as the period number that element is situated in.The electronic configuration of an element is related to its position on the periodic table. 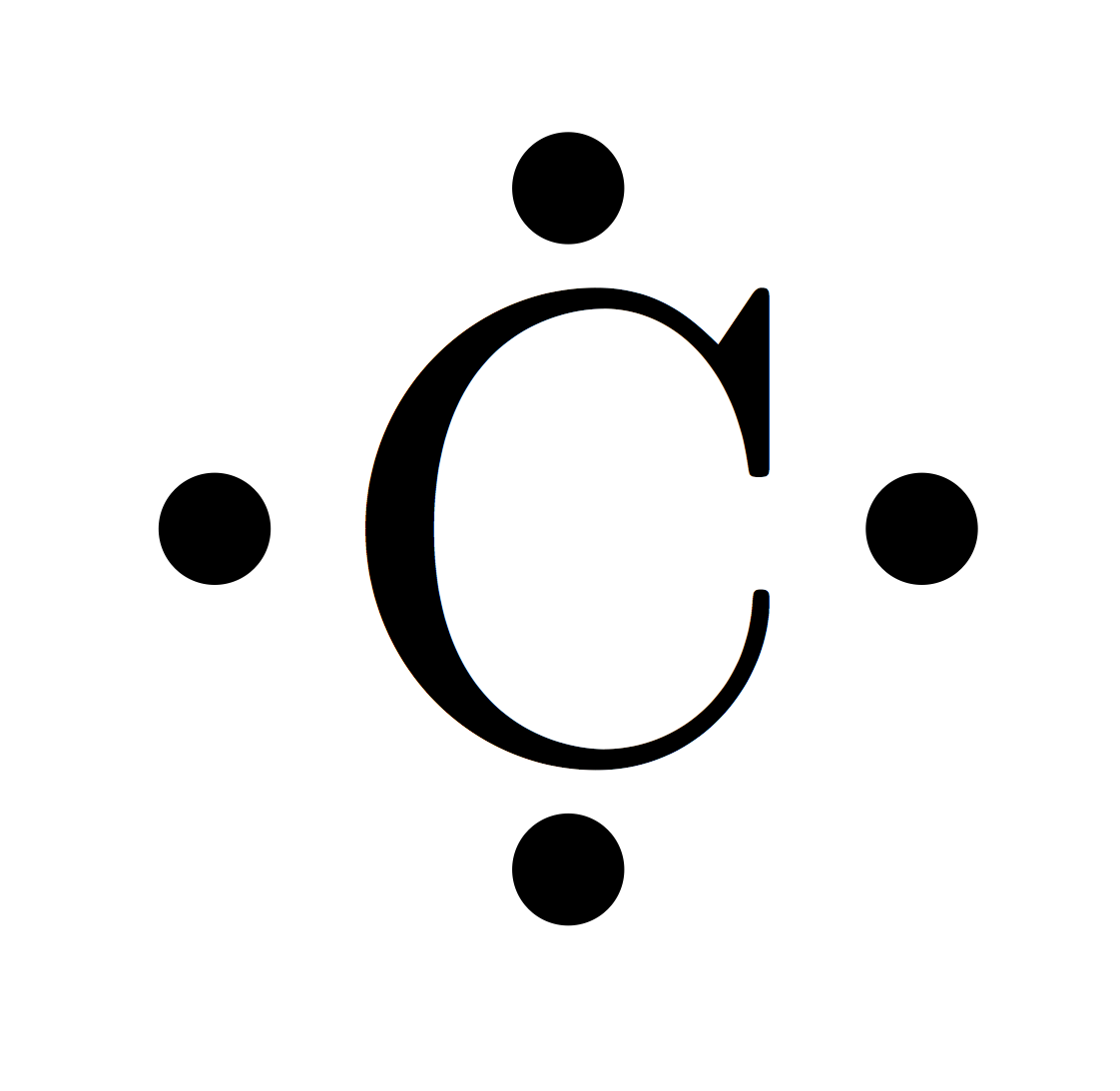
Electronic configurations and the periodic table
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
